BRAIN COMMISSURES
The brain commissures,
part of the three major types of connections of the neocortex (association,
projection,
and commissural fibers),
are those white matter tracts that cross the midline and connect the two cerebral hemispheres.
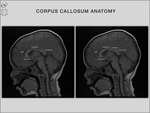
The largest commissure is the corpus callosum,
and thus it has the greatest contribution to the structural formation and shape of the brain.
It is divided into four well-known segments,
from anterior to posterior: rostrum,
genu,
body and splenium.
A fifth part is sometimes well seen and described – isthmus,
connecting the body and the splenium (Fig.
1).
The other two smaller interhemispheric fiber tracts are: 1.
anterior commissure (AC) – connecting the temporal lobes,
the olfactory areas and the amygdala and placed in front of the columns of the fornix and the anterior wall of the 3rd ventricle,
above the optic chiasm; 2.
hippocampal commissure (HC) – connecting the hippocampal formation and fornices and blended with the ventral surface of the callosal splenium [4].
Those three brain commissures develop almost simultaneously,
navigated by complex cellular and molecular relations,
and consequently,
anomalies associated with the interruption of this process are rarely isolated.
When the mechanisms regulating the commissural fibers formation and guidance fail,
pathological formation of one or more commissures occurs.
Callosal anomalies in most of the cases are accompanied by hippocampal commissure agenesis and in 50% by anterior commissure agenesis or hypoplasia [1,
2].
CALLOSAL ANATOMY
The corpus callosum has four segments,
but can be subdivided into two major parts: the anterior one,
consisting of the rostrum,
genu,
and body and the posterior one of splenium,
with a place of fusion between them – isthmus (Fig.
1).
Each callosal segment connects specific areas from the brain hemispheres,
plays a specific role in the transfer of information and therefore facilitates the coordination between them [1].
The rostrum is the most anterior-inferior callosal part,
forms the floor of the anterior horn of lateral ventricles and connects the fronto-basal orbital surfaces of the lobes.
The genu is the second part that is curved,
lies behind the frontal lobe,
and is formed of fibers that connect the prefrontal cortex and anterior cingulate area.
The trunk (body) is the main part of CC which connects the precentral motor cortex,
insula and cingulate gyri and forms the roof of the central part of lateral ventricles.
The isthmus is the thinner area between body and splenium and connects pre- and postcentral sensory-motor gyri,
as well as primary auditory areas.
The splenium is the most posterior massive part and connects the posterior parietal,
infero-medial temporal (including posterior cingulate) and medial occipital lobes [1,
5] (Fig.
2).
EMBRYOLOGY
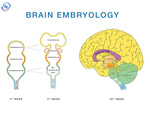
In the embryo (period from 2nd to 9th gestational weeks – g.w.) after the closure of the neuropore at 24-28th gestational days,
the neural tube starts to form and to differentiate into the three primary encephalic vesicles: prosencephalon (forebrain),
mesencephalon (midbrain) and rhombencephalon (hindbrain).
The prosecephalon is later divided into two parts: telecephalon and diencephalon (Fig.
3).
It is presumed that all telencephalic commissures cross the midline in a specific time through a distinct anatomical region – the commissural plate,
which serves as a zone for axonal passage and provides a glial pathway to guide commissural axons [3,
6].
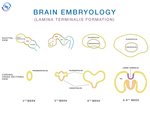
The telencephalon consists of two symmetric lateral evaginations – the cerebral hemispheres,
which at 6-8 g.w.
become separated by lamina terminalis at the rostral midline (Fig.
4).
The dorso-superior part of lamina terminalis becomes thicker and transforms into lamina reuiniens,
which ventral part later develops into area septalis.
As the hemispheric vesicles expand bilaterally,
the dorsal part of lamina reuiniens begins to fold into the median line,
its sides come closer and form the massa commissuralis [6,
7].
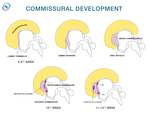
The interhemispheric commissures develop sequentially from two sites: at 10 g.w.
the anterior commissure from area septalis of lamina reuniens; and the hippocampal commissure and the corpus callosum later and dorsally from massa commissuralis (by interhemispheric fusion) (Fig.
5).
The earliest callosal fibers,
forming the genu and guided by pioneering cingulate axons,
are considered to appear and penetrate the massa commissuralis at 11 to 12 g.w.
[2,
6].
The callosal plate is well-defined in the fetus at 12-13 gestational weeks.
It is widely assumed that the embryological growth of the corpus callosum follows roughly anterior to posterior path,
beginning with the genu,
followed by the body,
splenium and completing with the rostrum at 20 g.w. [6].
A more detailed developmental sequence that was proposed is: posterior part of genu,
anterior callosal body,
anterior portion of genu at the same time as the posterior callosal body,
splenium and rostrum [8,
9].
However,
several studies exist which do not support these developmental theories,
offering bidirectional growth after the initial formation of a part of the body [3,
7].
After the characteristic callosal shape with its four parts is achieved at 20 g.w.,
active axonal growth in the CC continues up to 30 g.w.,
initially by addition of fibers and later by myelination [1,
2].
The formation of the corpus callosum can be divided into three main stages:
1) commissuration,
when fibers expand between the two telencephalic vesicles;
2) growth,
when each callosal part is created;
3) maturation,
pre- and postnatal axonal addition,
elimination and myelination.
In newborns the myelination of the CC and its final volume are not yet completed.
The thickness of the corpus callosum increases significantly throughout childhood and adolescence and reaches the target thickness at the age of 6 – 9 years.
Growth in the anterior sections is most noticeable in the first years of life,
while posterior splenial growth predominates later [1,
3].
CALLOSAL ANOMALIES
The corpus callosum passes through unique and complex developmental steps which can be divided into: neurogenesis,
midline patterning,
neuronal migration and specification,
axon guidance,
and post-guidance development.
Various environmental and genetic causes can interrupt the developmental process of the CC in different stages,
leading to congenital malformations associated with particular alterations in the brain structure and spectrum of neuropsychological deficits [2].
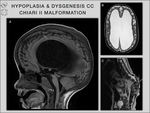
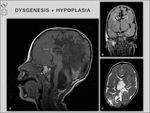
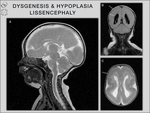
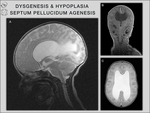
There are several major types of callosal anomalies: agenesis – complete absence of the corpus callosum; dysgenesis – partial formation,
with missing segments; hypoplasia – preserved integrity and shape,
but smaller size [1,
3,
6].
The specific congenital anomalies of the brain,
and particularly of the corpus callosum,
occur in a particular time of the embryological developent (Fig.
6).
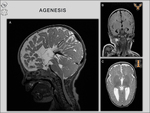
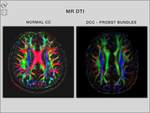
1/ Agenesis: Agenesis of the corpus callosum (ACC) is most commonly associated with axons that fail to cross the midline and form atypical longitudinal fibers – Probst bundles and cause anomalous ventricle appearance (Fig.
7).
However,
in small number of cases axons do not form at all,
thus no Probst bundles are present.
ACC is a heterogeneous condition that can result from disruption of numerous developmental steps – from early midline telencephalic formation to post-guidance alterations [3].
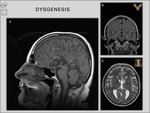
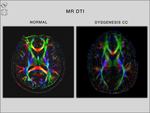
2/ Dysgenesis: Typical dysgenesis of the corpus callosum (DCC) consists in the presence of the earlier-formed segments (genu,
body) and absence of the later-formed ones (splenium,
rostrum) (Fig.
8).
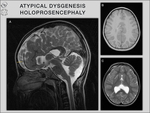
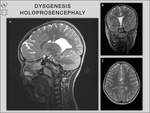
In some patients with holoprosencephaly (semilobar or middle interhemispheric variant),
condition that results from disruption of the prosepncephalic cleavage between 7-8 g.w.,
an atypical DCC can be observed.
In such cases with interhemispheric fusion,
due to lack of induction of the normal commissural plate,
the normal sequence of callosal formation is not followed and therefore presence of the latest-formed segments and absence of the genu and part of body is noticed [6,
8] (Fig.
9).
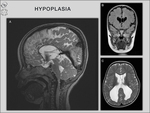
3/ Hypoplasia: In cases with hypoplasia,
the corpus callosum is thinner than general,
but has a normal anterior–posterior extent [3] (Fig.
10).
MR IMAGING
The exact anatomy of the corpus callosum,
all types of its malformations,
as well as additional brain anomalies,
can be evaluated accurately with MRI of the brain.
The typical adult signal intensity of the corpus callosum is observed when its complete myelination (from posterior to anterior – genu last) is achieved after 1st postnatal year – hyperintense on T1-weighted images and hypointense on T2-weighted images [1,
4].
MR diffusion tensor imaging (DTI),
a noninvasive method based on the analysis of water diffusion in brain,
accurately depicts the normal callosal interhemispheric connections,
but also the abnormal alignment of the white matter tracts.
It reveals the normal callosal white matter fibers that interconnect homotopic areas of both hemispheres.
In cases with callosal maldevelopment,
this technique represents precisely the disturbed and re-routed connections,
such as the bilateral aberrant antero-dorsal running Probst bundles [2,
3] (Fig.
11,
12).
In studies of individuals with partial callosal agenesis,
DTI revealed extremely variable callosal connectivity,
with many heterotopic tracts not seen in healthy subjects,
for example ‘sigmoid bundles’ connecting the anterior frontal lobe with the contralateral parieto-occipital [3,
10].
Corpus callosum contributes mainly to the shape and size of the brain ventricles and as a result of its malformation specific changes in their structure,
size and position in MRI are found.
In callosal agenesis,
the heterotopically positioned Probst bundles,
oriented longitudinally and parallelly to the interhemispheric fissure and seen as hyper- at T1 weighted imaging and hypointense signal at T2WI,
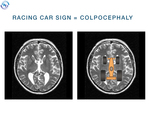
cause parallel orientation of the lateral ventricles and dilatation of the atria and occipital horns (colpocephaly) – ‘tear drop’ appearance on axial scans [2].
In axial MRI plane,
these characteristic ventricular changes are described as “racing car sign”,
due to the resemblance of a race car (Fig.
13).
Moreover,
the commonly associated hypoplastic hippocampal formation leads to dilatation of the temporal ventricular horns.
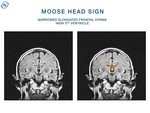
Another related brain alteration is the position of the third ventricle,
as it may be abnormally dilated and bulging upward,
giving the appearance of a cyst,
or it may communicate with the interhemispheric cistern.
This in coronary plane,
along with the malrotated or absent cingulate gyrus that leads to narrowed elongated frontal horns,
gives the sign of “moose head” or also called ”Viking helmet” (Fig.
14).
In some cases,
in sagittal plane,
abnormally formed gyri and sulci are found,
converging radially and medially toward the 3rd ventricle – "sunray appearance" [1,
6] (Fig.
7).
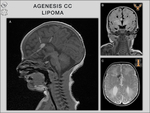
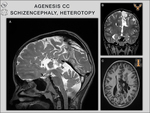
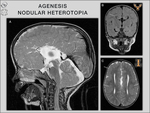
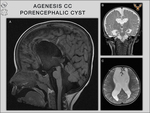
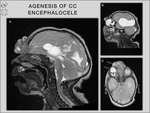
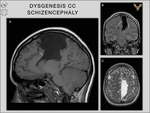
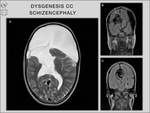
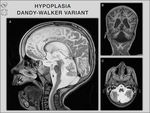
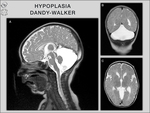
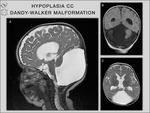
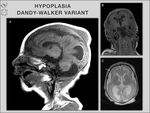
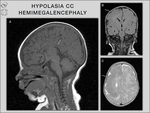
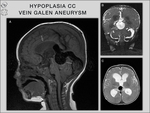
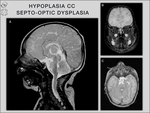
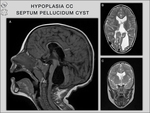
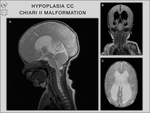
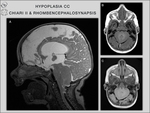
Callosal anomalies can be associated with many other brain malformations,
from which we have encountered:
lipoma,
schizencephaly,
nodular heterotopia,
polymicrogyria,
porencephalic cyst,
encephalocele,
holoprosencephaly,
Dandy-Walker malformation,
hemimegalencephaly,
hydrocephalus due to vein of Galen aneurysm,
septo-optic dysplasia,
septum pellucidum cyst,
Chiari II malformation,
rhombencephalosynapsis,
lissencephaly and septum pellucidum agenesis (Fig.
15-36).